Quattroflow™, part of PSG®, a Dover company and a leading brand of positive displacement quaternary diaphragm pumps, is pleased to announce that its Single-Use Quaternary (Four-Piston) Diaphragm Pumps have been designed to help eliminate particle contamination in biopharmaceutical applications. This makes Quattroflow pumps ideally suited for optimizing the handling of expensive and/or sensitive liquids often found in these types of applications.
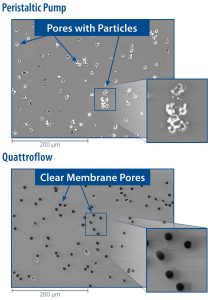 To help verify the ability of its pumps to minimize generation, Quattroflow recently commissioned a third-party test with a hypothesis that the gentle working principle of the Quattroflow quaternary pump minimizes component stress and, thus, the possible generation of particles. The test featured a comparison between a Quattroflow QF150SU model pump and a peristaltic pump that was using a pharma-grade pumping hose. Both pumps performed an eight-hour continuous recirculation of a liquid through a 12μm filter at approximately 100 L/hr (26.4 gph).
To help verify the ability of its pumps to minimize generation, Quattroflow recently commissioned a third-party test with a hypothesis that the gentle working principle of the Quattroflow quaternary pump minimizes component stress and, thus, the possible generation of particles. The test featured a comparison between a Quattroflow QF150SU model pump and a peristaltic pump that was using a pharma-grade pumping hose. Both pumps performed an eight-hour continuous recirculation of a liquid through a 12μm filter at approximately 100 L/hr (26.4 gph).
After the test was completed, an examination of the filter membranes showed that 2 Mio particles with sizes between 6.1 and 12.7μm were generated during the operation of the peristaltic pump. Conversely, no identifiable particles were generated during the operation of the Quattroflow pump just clear filter membrane pores. These test results show that Quattroflow pumps help eliminate particle generation and subsequent contamination of the product and are suitable for optimizing the handling of expensive and/or sensitive liquids in biopharmaceutical-manufacturing operations. A flyer of these test results has been created that can be downloaded from the Quattroflow Single-Use Pumps web page or by clicking here.
In addition to minimizing product particle contamination, the implementation of Quattroflow single-use pumps also  create a quicker production process for biopharmaceutical manufacturers, as well as one that delivers the required levels of product purity and sterility with no corresponding harm to the product and no chance for costly cross-batch or cross-product contamination. Other features and benefits that make Quattroflow single-use pumps ideal for use in biopharmaceutical applications include reduced downtime, low pulsation, consistent flow rate, quiet operation, superior containment, self-priming and dry-running capabilities, low heat input, high turn-up/down ratio and optional injection-molded PE pump chamber. Quattroflow single-use pumps are available in four sizes, covering a performance range from 1 to 5,000 L/hr (0.26 to 1,321 gph).
create a quicker production process for biopharmaceutical manufacturers, as well as one that delivers the required levels of product purity and sterility with no corresponding harm to the product and no chance for costly cross-batch or cross-product contamination. Other features and benefits that make Quattroflow single-use pumps ideal for use in biopharmaceutical applications include reduced downtime, low pulsation, consistent flow rate, quiet operation, superior containment, self-priming and dry-running capabilities, low heat input, high turn-up/down ratio and optional injection-molded PE pump chamber. Quattroflow single-use pumps are available in four sizes, covering a performance range from 1 to 5,000 L/hr (0.26 to 1,321 gph).
For more information on Quattroflow, please visit quattroflow.com.
About Quattroflow:
Quattroflow™ is a leading brand of Almatec® Maschinenbau GmbH, Kamp-Lintfort, Germany, which is part of PSG®, a Dover company. Quattroflow pumps offer a four-piston diaphragm technology and primarily serve industries such as pharmaceutical and biopharmaceutical that require gentle displacement, reliability, product safety, purity and cleanability. For more information on Quattroflow and its complete family of four-piston pumps, please visit quattroflow.com. For more information on PSG and any of its leading brands, please visit psgdover.com.




